A bright idea: turning food waste into LEDs
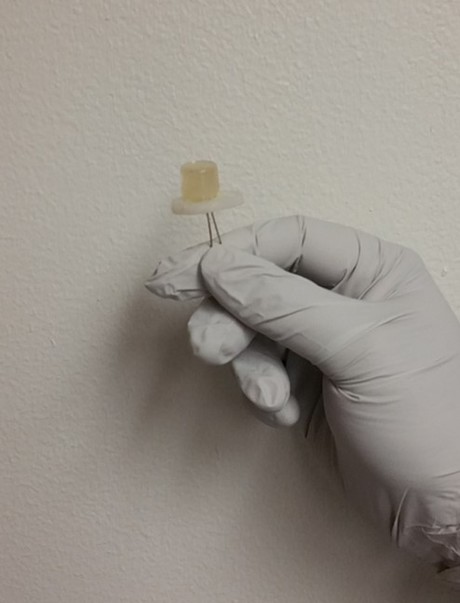
Light-emitting diodes (LEDs) have been an efficient alternative to fluorescent and incandescent bulbs for several years. Now, University of Utah researchers have found a way to make LEDs even more sustainable than they already are — by creating them from food and beverage waste.
LEDs can be produced by using quantum dots (QDs), or tiny crystals that have luminescent properties, to produce light. QDs can be made with numerous materials, some of which are rare and expensive to synthesise — and even potentially harmful to dispose of.
Research over the past 10 years has focused on using carbon dots (CDs), QDs made of carbon, to create LEDs instead. CDs have lower toxicity and better biocompatibility than other types of QDs, meaning they can be used in a broader variety of applications. But University of Utah Professors Prashant Sarswat and Michael Free have gone one step further, successfully turning food waste into CDs and, subsequently, LEDs.
Sarswat and Free employed a solvothermal synthesis, in which the waste was placed into a solvent under pressure and high temperature until CDs were formed. In this experiment, the researchers used soft drinks and pieces of bread and tortilla. The food and beverage waste were each placed in a solvent and heated both directly and indirectly for anywhere from 30 to 90 min.
After successfully finding traces of CDs from the synthesis, Sarswat and Free proceeded to illuminate the CDs to monitor their formation and colour. The pair also employed four other tests — Fourier transform infrared spectroscopy, X-ray photoelectron spectroscopy, and Raman and AFM imaging — to determine the CDs’ various optical and material properties.
“Synthesising and characterising CDs derived from waste is a very challenging task,” said Sarswat. “We essentially have to determine the size of dots which are only 20 nanometres or smaller in diameter, so we have to run multiple tests to be sure CDs are present and to determine what optical properties they possess.”
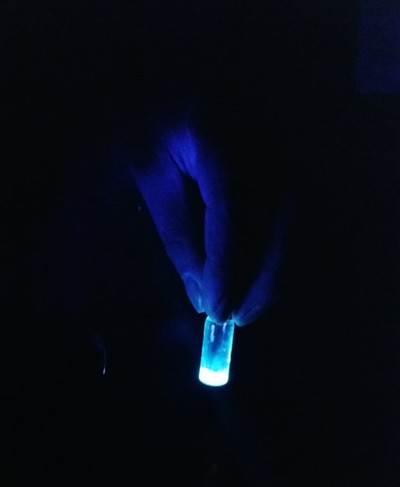
The various tests first measured the size of the CDs, which correlates with the intensity of the dots’ colour and brightness. They then determined which carbon source produced the best CDs, with sucrose and D-fructose dissolved in soft drinks found to be the most effective sources. Finally, the CDs were suspended in epoxy resins, heated and hardened to solidify them for practical use in LEDs.
Not only do the researchers’ LEDs utilise products that would otherwise decompose, they will also reduce potentially harmful waste from the LEDs themselves. One of the most common sources of QDs is cadmium selenide, a compound comprising two toxic elements. As explained by Free, “QDs derived from food and beverage waste are not based on common toxic elements such as cadmium and selenium, which makes their processing and disposal more environmentally friendly than it is for most other QDs.”
Cadmium selenide is also expensive, with one website listing a price of $529 for 25 mL of the compound. Food and beverage waste, on the other hand, is both readily available — 31% of food produced in 2014 was not available for human consumption, according to the US Department of Agriculture — and essentially free.

The results of Sarswat and Free’s study have been published in Physical Chemistry Chemical Physics, a journal of the Royal Society of Chemistry. The researchers hope to continue studying the LEDs produced from food and beverage waste for stability and long-term performance; the ultimate goal, according to Sarswat, “is to do this on a mass scale and to use these LEDs in everyday devices”.
Why data centres must rethink water use
In a world of increasing freshwater scarcity, we must balance digital innovation with responsible...
More could be done for sustainability in Budget, say industry groups
Industry and environmental leaders have provided strong reviews of the federal Budget after green...
Office tower cuts HVAC energy use by nearly 50%
The building has cut almost half of the HVAC electricity use and 5% of gas consumption annually,...










